When Rutherford was five he moved to Foxhill and attended Foxhill School. 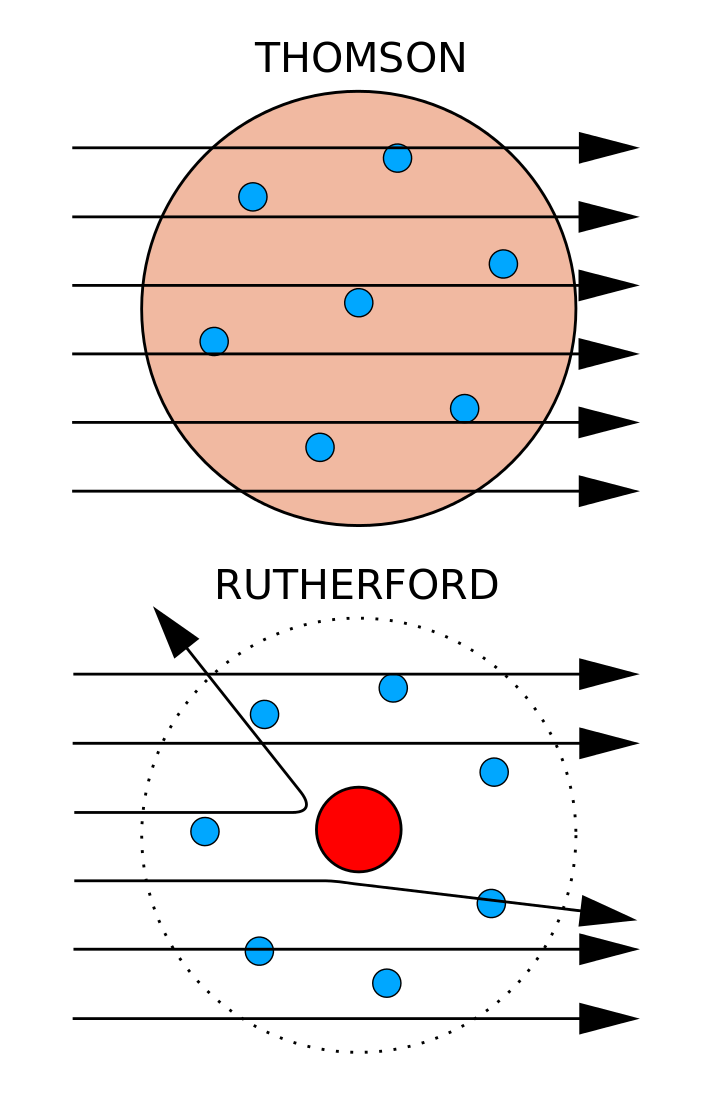
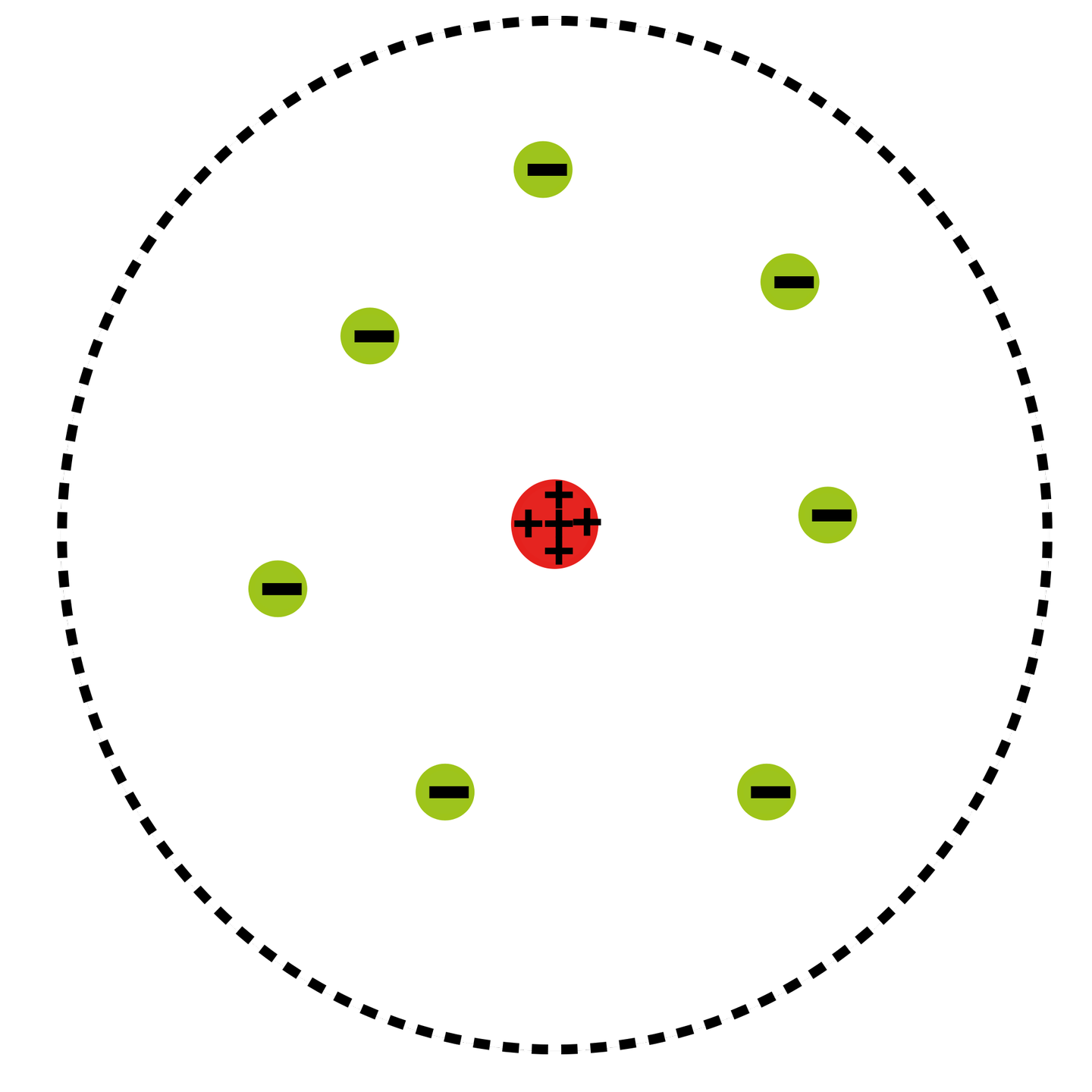
Rutherford's birth certificate was mistakenly written as 'Earnest'. He was the fourth of twelve children of James Rutherford, an immigrant farmer and mechanic from Perth, Scotland, and his wife Martha Thompson, a schoolteacher from Hornchurch, England. The chemical element rutherfordium ( 104Rf) was named after him in 1997.Įrnest Rutherford was born on 30 August 1871 in Brightwater, a town near Nelson, New Zealand. After his death in 1937, he was buried in Westminster Abbey near Charles Darwin and Isaac Newton. In honour of his scientific advancements, Rutherford was recognised as a Baron in the peerages of New Zealand and Britain. In the same year, the first controlled experiment to split the nucleus was performed by John Cockcroft and Ernest Walton, working under his direction. Under his leadership, the neutron was discovered by James Chadwick in 1932. Rutherford became Director of the Cavendish Laboratory at the University of Cambridge in 1919. His other achievements include advancing the fields of radio communications and ultrasound technology. He is also credited with developing the atomic numbering system alongside Henry Moseley. As a result, he discovered the emission of a subatomic particle which he initially called the "hydrogen atom", but later (more accurately) named the proton. In 1917, he performed the first artificially-induced nuclear reaction by conducting experiments where nitrogen nuclei were bombarded with alpha particles. This was done through his discovery and interpretation of Rutherford scattering during the gold foil experiment performed by Hans Geiger and Ernest Marsden, resulting in his conception of the Rutherford model of the atom. In 1911, he theorized that atoms have their charge concentrated in a very small nucleus. Together with Thomas Royds, Rutherford is credited with proving that alpha radiation is composed of helium nuclei. Rutherford's discoveries include the concept of radioactive half-life, the radioactive element radon, and the differentiation and naming of alpha and beta radiation. In 1908, he was awarded the Nobel Prize in Chemistry "for his investigations into the disintegration of the elements, and the chemistry of radioactive substances." He was the first Oceanian Nobel laureate, and the first to perform the awarded work in Canada. Rutherford has been described as "the father of nuclear physics", and "the greatest experimentalist since Michael Faraday". He concluded that all of the positive charge and the majority of the mass of the atom must be concentrated in a very small space in the atom's interior, which he called the nucleus.Ernest Rutherford, 1st Baron Rutherford of Nelson, OM, PRS, HonFRSE (30 August 1871 – 19 October 1937) was a New Zealand physicist who was a pioneering researcher in both atomic and nuclear physics. In contrast, the particles that were highly deflected must have experienced a tremendously powerful force within the atom. Because the vast majority of the alpha particles had passed through the gold, he reasoned that most of the atom was empty space. Rutherford needed to come up with an entirely new model of the atom in order to explain his results. In a famous quote, Rutherford exclaimed that it was "as if you had fired a 15-inch shell at a piece of tissue and it came back and hit you." No prior knowledge had prepared them for this discovery. Some were even redirected back toward the source. Surprisingly, while most of the alpha particles were indeed not deflected, a very small percentage (about 1 in 8000 particles) bounced off the gold foil at very large angles. Rutherford found that a small percentage of alpha particles were deflected at large angles, which could be explained by an atom with a very small, dense, positively-charged nucleus at its center (bottom).Īccording to the accepted atomic model, in which an atom's mass and charge are uniformly distributed throughout the atom, the scientists expected that all of the alpha particles would pass through the gold foil with only a slight deflection or none at all. (B) According to the plum pudding model (top), all of the alpha particles should have passed through the gold foil with little or no deflection. \): (A) The experimental setup for Rutherford's gold foil experiment: A radioactive element that emitted alpha particles was directed toward a thin sheet of gold foil that was surrounded by a screen which would allow detection of the deflected particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
